Physical Activity Reduces Stress-Related Brain Activity to Lower Cardiovascular Disease Risk
New research indicates that physical activity lowers cardiovascular disease risk in part by reducing stress-related signaling in the brain.
Department of Medicine
Contact Information
CNY 149-8
149 13th Street
Charlestown,
MA
02129
Phone: 617-726-5663
Fax: 617-726-5671
Email: aarnaout1@mgh.harvard.edu
Hours:
Monday–Friday, 8:00 am–6:00 pm
Grants Manager: Diane DeAngelis
Phone: 617-724-1551
Email: ddeangelis2@mgh.harvard.edu
Laboratory Manager: Zhiping Ding
Phone: 617-724-9875
Email: zding@partners.org
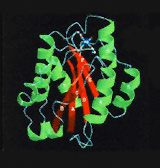
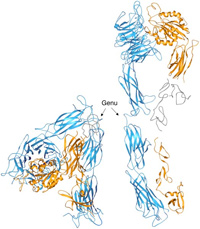
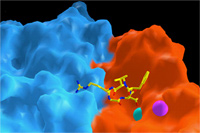
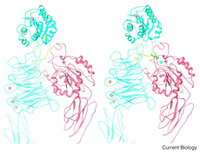
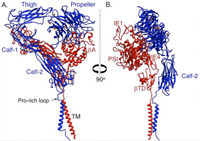
A major research goal of the Arnaout Laboratory is to elucidate the structure and function of integrins, cell adhesion receptors that play vital roles in normal physiology and disease and use the derived information in structure-based design of new and safer anti-integrin drugs targeting heart disease, fibrosis, and cancer.
Other research interests include elucidating mechanisms underlying cyst formation in Autosomal Dominant Polycystic Kidney Disease, transcriptional regulation of hematopoiesis, mechanisms of kidney regeneration, and design of microfluidic dialysis devices.
 |
|
 |
|
 |
|
 |
|
 |
|
 |
|
 |
|
If you are interested in applying for a postdoctoral position, or are a Harvard PhD student interested in a laboratory rotation, please e-mail your CV (for student and postdoctoral) and reference letters (for postdoctoral) to: aarnaout1@mgh.harvard.edu
See a list of publications from this investigator.
Every day, our clinicians and scientists chart new terrain in biomedical research to treat and prevent human disease and advance patient care.
Your support of Nephrology helps us provide the best possible care today and expand what will be possible tomorrow.
We offer innovative, high-quality medical care, trains future medical leaders, and produces research that advances science and improves care.
New research indicates that physical activity lowers cardiovascular disease risk in part by reducing stress-related signaling in the brain.
Results warrant additional studies into the health benefits of aspirin for patients with metabolic dysfunction–associated steatotic liver disease.
Researchers have identified numerous genetic clusters that may help explain why people with type 2 diabetes have different clinical presentations.
We reached out to investigators at Massachusetts General Hospital and asked them to give us one science-backed action people can take to improve their heart health.
In a nationally representative of US adults with self-reported chronic liver disease (CLD), approximately 308,000 (6%) identified delaying medical care due to transportation barriers.
In this 12-month, placebo-controlled, randomized clinical trial of 26 well-treated persons with HIV without known CVD, eplerenone was associated with a significant reduction in arterial inflammation.
The Division of Nephrology at Massachusetts General Hospital is a leading provider of services for patients with kidney disease, including diagnosis and management of kidney diseases and medical management of renal transplantation.