Cobbold Lab


Cobbold Lab
Mark Cobbold, MRCP, PhD
Associate Professor of Medicine
Massachusetts General Cancer Center
Harvard Medical School
Program Affiliations
Center for Cancer Research
Explore the Cobbold Lab
Research Summary
The Cobbold laboratory focuses on understanding how the healthy human immune response is able to recognize and target cancerous cells, and when it fails, how it could be strengthened to recognize this endogenous threat. Our immune system faces a challenge in targeting cancerous cells as they are not “foreign” yet subtle changes in the cellular proteins exist that nevertheless allow our immune cells (T-cells) to detect them. Cancer cells modify internal proteins in different ways to healthy cells, a process fundamental to a cell becoming cancerous. These abnormal modifications can be recognized by T-cells and we believe play a role in protecting us against cancer.
In patients with cancer, this immunity is often lost and therefore new approaches may be needed. We are developing therapeutics that mobilize existing potent immune responses to instead tackle cancer. These immune-based therapies make cancerous cells appear virally infected in the hope that our own anti-viral immunity could step in to fill the void.
Research Projects
Tumor Neoantigens
Recently, new therapies such as checkpoint blockade therapies have revealed potent underlying anti-tumor immunity in patients with many types of cancer. The nature of the tumor antigens targeted by endogenous immunity is still being characterized, but “altered-self” antigens (neoantigens) are thought to play a dominant role. Neoantigens can be generated through genetic mutations that lead to changes in protein sequence (so called “mutational neoantigens”), but these types of antigens are typically unique to each tumor and thus any approach targeting these would need to be in a form of a personalized therapeutic.
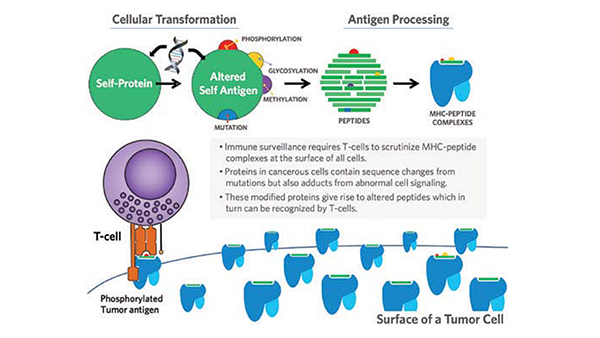
We have identified posttranslational modifications (phosphorylation, glycosylation and methylation) as additional mechanisms whereby proteins can be abnormally modified allowing immune recognition. Thus, aberrant signaling renders phosphate, methyl-, or N-GlcNAc protein adducts as pathophysiological haptens (Cancer Immunol Res. 2017). These antigens are abundantly displayed on the surface of cancer cells bound to MHC class-I molecules and we have identified over 1,000 phosphorylated peptides found on primary tumors. In contrast to classical neoantigens, the surface-display of posttranslationally modified neoantigens is shared between many tumor types potentially affording a broader targeting approach.
This class of tumor antigens exhibits interesting MHC binding with, for example, increased affinity for MHC molecules over non-phosphorylated counterparts. Moreover, structural studies reveal an MHC surface binding pocket facilitating the presentation of the phosphate groups to T-cells (Nature Immunology 2008). This recognition pathway may have evolved to allow T-cells to scrutinize underlying cellular signal transduction pathways directly linking adaptive immunity with protection against fundamental oncogenic processes.
We have shown the existence of memory-like T-cells in healthy individuals that recognizes leukemia-associated phosphopeptide antigens, yet this immunity is lost in patients with leukemia (Science Translational Medicine 2013). Immunotherapies, such as stem cell transplantation where healthy donor immunity is transferred to patients, restore this immunity providing some evidence for its potential utility. Thus, posttranslationally modified antigens represent a new class of neoantigens, but their importance in cancer remains to be determined.
T-cell Activating Biotherapeutics
When detected in the blood, immunity against cancer is known to be weak, exhibiting low frequencies, and of compromised function. This contrasts starkly against immunity toward persistent viruses such as human cytomegalovirus (CMV) or Epstein-Barr virus (EBV) which typically occupy exceptionally high frequencies in both blood and tissues. Moreover, it is known that CMV immunity increases with age and is preserved in patients with cancer, thus making it ideal to harness against tumors.
We have advanced the notion of altered-self and developed a technology that is able to manipulate the types of antigens present on the surface of tumor cells. We utilized dominant CMV antigens (we term “über-antigens”) to develop peptide payloads that could be conjugated to tumor-targeting antibodies. These complexes allow surface release of the über-antigens with subsequent passive loading into empty MHC class-I molecules by exploiting proteases expressed by cancer and engineering antigens that can be processed by these proteases. Thus, this approach circumvents the requirement in the host of potent anti-cancer immunity which is lacking.
Most recently, we have taken the approach of engineering antibodies that cooperatively engage with immune effector cells to target cancer cells with ultra-specificity through logic gated activation.
Publications
Selected Publications
Malaker SA, Penny SA, Steadman LG, Myers PT, Loke JC, Raghavan M, Bai DL, Shabanowitz J, Hunt DF, Cobbold M. Identification of Glycopeptides as Posttranslationally Modified Neoantigens in Leukemia. Cancer Immunol Res. 2017 May;5(5):376-384.
Wong GK, Millar D, Penny S, Heather JM, Mistry P, Buettner N, Bryon J, Huissoon AP & Cobbold M. Accelerated Loss of TCR Repertoire Diversity in Common Variable Immunodeficiency. J. Immunol.2016 Sep 1;197(5):1642-9. PMID: 27481850.
Cobbold M*, De La Peña H, Norris A, Polefrone JM, Qian J, English AM, Cummings KL, Penny S, Turner JE, Cottine J, Abelin JG, Malaker SA, Zarling AL, Huang HW, Goodyear O, Freeman SD, Shabanowitz J, Pratt G, Craddock C, Williams ME, Hunt DF, Engelhard VH. MHC class I-associated phosphopeptides are the targets of memory-like immunity in leukemia. Sci Transl Med. 2013 Sep 18;5(203):203ra125.
Mohammed F*, Cobbold M*, Zarling AL, Salim M, Barrett-Wilt GA, Shabanowitz J, Hunt DF, Engelhard VH, Willcox BE. Phosphorylation-dependent interaction between antigenic peptides and MHC class I: a molecular basis for the presentation of transformed self. Nat Immunol. 2008 Nov;9(11):1236-43. doi: 10.1038/ni.1660. Epub 2008 Oct 5. PubMed PMID: 18836451.
* Joint first author
*Corresponding Author
†Co-first authorship
Research Image
Model for generation of modified neoantigens as targets for cancer immunosurveillance.
Our Researchers
Mark Cobbold, MRCP, PhD
Principal InvestigatorGroup Members
- Jamie Heather, PhD
- David G. Millar, PhD
- Sergio Nunez
- Sean Sepulveda
- Feng Shi, PhD
- Yifang Ivana Shui, PhD
- Li Wan
- Songfa Zhang, PhD
Krantz Family Center for Cancer Research
The scientific engine for discovery for the Mass General Brigham Cancer Institute.
Support the Krantz Family Center for Cancer Research
When you support us you are enabling discoveries that will lead to effective new weapons in the battle against cancer.
We are home to more than 1,000 active clinical trials per year.
Ensuring that our patients have access to pioneering therapies.
