Krantz Family Center for Cancer Research
Hock Lab
Contact Information
Hock Lab
Simches Building, Mail code CPZN 4200
185 Cambridge Street, 4th floor
Boston,
MA
02114
Phone: 617-643-3145
Email: hhock@partners.org
Hanno Hock, MD, PhD
Assistant Professor of Medicine
Harvard Medical School
Brant Carleton Endowed Chair in Acute Myeloid Leukemia Research
Program Affiliations
- Krantz Family Center for Cancer Research
- Hematology & Medical Oncology
- Center for Regenerative Medicine
Explore the Hock Lab
Research Summary
The Hock laboratory explores the molecular basis of blood cell formation and the pathogenesis of leukemia and lymphoma. Specifically, we study the transcription factors that regulate gene activity during normal blood cell development and how the transcriptional apparatus goes awry in cancer. For example, we have developed important insights into a network of transcription factors that help maintain blood stem cells in the bone marrow; this work could lead to new strategies for increasing the yield of stem cells for bone marrow transplantation. Another project in our laboratory focuses on deciphering the multistep process that leads to lymphoblastic leukemia of childhood, with the goal of identifying new drug targets for this devastating disease. Finally, we are interested in how DNA packaging affects the interaction between genes and transcription factors, especially with regard to oncogenes and tumor suppressor genes important in human cancer.
Research Projects
Our laboratory is interested in the molecular control of normal and malignant stem cells with an emphasis on the hematopoietic system. Blood cells need to be continuously replenished by a small population of hematopoietic stem cells (HSCs) that have the capacity to both self-renew and mature stepwise into all known blood lineages. HSCs are also the ancestors of leukemia and lymphoma cells. As HSCs mature, they undergo successive changes in gene expression. The transcriptional apparatus must ensure that genes specific to immature cells are repressed as differentiation proceeds, while genes that are necessary for mature cells become activated. This activating and inactivating of genes is achieved by cooperative action of a variety of lineage-specific and general transcription factors and the complex molecular machinery that regulates the accessibility of different regions of the genome in chromatin. We investigate how transcription factors establish differentiation-specific transcriptional programs and how such programs can become derailed in cancer, leukemia and lymphoma.
Transcriptional control of normal and malignant hematopoietic stem cells in the adult bone marrow
Hematopoiesis in the bone marrow emanates HSCs. We are studying the basic biology of HSCs. Specifically we explore how a network of transcription factors that includes Tel- Etv6, Gfi1, Gfi1b and Gata2 maintains HSCs in the bone marrow (Hock et al. 2004, Genes & Development; Hock et al. 2004, Nature). The goal is to exploit the biology of transcriptional regulation of HSCs to maintain, expand, and possibly even generate HSCs ex vivo so that more patients will have the option of bone marrow transplantation. In a closely related effort, we are exploring the molecular programs of stem cells in leukemia and lymphoma to identify differences in their molecular regulation compared with normal HSCs. Such differences may allow us to specifically target tumor stem cells while sparing normal blood formation.
Deciphering the molecular events leading to acute lymphoblastic leukemia of childhood
About one in 2000 children develops this catastrophic illness, most often with a t(12;21) translocation. Despite very aggressive treatments, not all children can be cured, and some suffer from longterm side effects of their therapy. Rational development of more specific, less toxic treatments requires a precise understanding of the molecular mechanisms that cause the disease. We have discovered that TEL-AML1, the first hit in childhood leukemia, generates a preleukemic, latent lesion in HSCs. We are now exploring how additional genetic hits cooperate to derail normal blood development and generate leukemia. Deciphering the multistep pathogenesis of this entity is likely to serve as a paradigm for the development of other malignant diseases.
Exploration of novel epigenetic regulators in stem cells
Our understanding of how specialized cells of the body establish their identity by regulating access to genes continues to increase. For example, a large fraction of the genes active in brain cells are inactive in blood cells and, therefore, are stored in a very dense, inaccessible state. As most molecules involved in the regulation of gene accessibility have only recently been identified, studying their biology is likely to provide unique opportunities for the development of entirely novel therapies. We are investigating the utility of a group of proteins termed MBT-proteins, which is very important for condensing DNA and modifying histones. Evidence suggests that this protein family may play important roles in normal and malignant blood formation, but its precise functions remain poorly understood. Our laboratory has recently discovered an entirely novel, essential function of the family member L3mbtl2 in pluripotent stem cells.
Research Positions
Postdoctoral Research Fellow (1)
A postdoctoral Research Fellow position is available in the laboratory of Hanno Hock, MD, PhD, Massachusetts General Hospital, Harvard Medical School, for studying the transcriptional regulation of stem cells in normal hematopoiesis and leukemia using genetically engineered mice. The laboratory is located in the new Simches Research Building at the main campus of the Massachusetts General Hospital. The fellow will have simultaneous appointments at the Cancer Institute and Center for Regenerative Medicine of MGH and Harvard Medical School and will be affiliated with the Harvard Stem Cell Institute. Candidates are expected to have completed their PhD and/or MD degree (with significant prior research experience) and be highly motivated for a career in academic biomedical research.
Contact
Please email a brief cover letter and CV to:
Assistant Professor of Medicine
Massachusetts General Hospital
Harvard Medical School
Simches Research Building, CPZN 4200, 4th floor
Boston, MA 02114
E-mail: Hock.Hanno@mgh.harvard.edu
Publications
View a list of publications by researchers at the Hock Laboratory
Selected Publications
Nardi V, Ku N, Frigault MJ, Dubuc AM, Tsai HK, Amrein PC, Hobbs GS, Brunner AM, Narayan R, Burke ME, Foster J, Dal Cin P, Maus MV, Fathi AT, Hock H. Clinical response to larotrectinib in adult Philadelphia chromosome-like ALL with cryptic ETV6-NTRK3 rearrangement. Blood Adv. 2020;4(1):106-11.
Brumbaugh J, Kim IS, Ji F, Huebner AJ, Di Stefano B, Schwarz BA, Charlton J, Coffey A, Choi J, Walsh RM, Schindler JW, Anselmo A, Meissner A, Sadreyev RI, Bernstein BE, Hock H*, Hochedlinger K*. Inducible histone K-to-M mutations are dynamic tools to probe the physiological role of site-specific histone methylation in vitro and in vivo. Nat Cell Biol. 2019;21(11):1449-61.
Hock, H., and A. Shimamura. 2017. ETV6 in hematopoiesis and leukemia predisposition. Seminars in hematology 54:98-104. PMC5584538
Foudi A, Kramer DJ, Qin J, Ye D, Behlich AS, Mordecai S, Preffer FI, Amzallag A, Ramaswamy S, Hochedlinger K, Orkin SH and Hock H. Distinct, strict requirements for Gfi-1b in adult bone marrow red cell and platelet generation. J Exp Med 211, 909 – 927. 2014.
Qin J, Whyte WA, Anderssen E, Apostolou E, Chen H, Akbarian S, Bronson RT, Hochedlinger K, Ramaswamy S, Young RA, and Hock H. The Polycomb Group Protein L3mbtl2 Assembles an Atypical PRC1-family Complex with Essential Roles in Pluripotent Stem Cells and Early Development. Cell Stem Cell. 2012 . 11, 319-332, 2012.
Schindler JW, Van Buren D, Foudi A, Krejci O, Qin J, Orkin SH, Hock H. TEL-AML1 corrupts hematopoietic stem cells to persist in the bone marrow and initiate leukemia. Cell Stem Cell. 5(1):43-53, 2009 Jul 2.
*corresponding authorsResearch Image
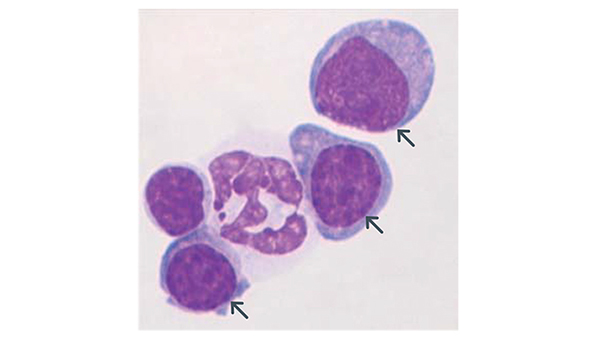
Dr. Hock's laboratory works on molecular mechanisms of normal differentiation and malignant transformation. The image shows normal blood cells and leukemic cells (arrows) from a novel experimental model generated in the lab.
Our Researchers
Hanno Hock, MD, PhD
Principal InvestigatorGroup Members
- Daniel Kramer
- Ondrej Krejci, PhD
- Ryan LeGraw
Krantz Family Center for Cancer Research
The scientific engine for discovery for the Mass General Brigham Cancer Institute.
Support the Krantz Family Center for Cancer Research
When you support us you are enabling discoveries that will lead to effective new weapons in the battle against cancer.
