Goldstein Lab: Allan Goldstein, MD
Contact Information
Allan Goldstein Lab
Charles River Park Plaza North
185 Cambridge Street
Boston,
MA
02114
Phone: 617-726-0270
Fax: 617-726-2167
Email: agoldstein@partners.org
Research Topics
Stem Cell Therapy for Hirschsprung Disease and Other Neurointestinal Diseases
One of our primary areas of investigation is to develop neuronal stem cell transplantation as a novel treatment for neurointestinal diseases, such as Hirschsprung disease. We have established methods to isolate neuronal stem cells from the intestines of mice and human patients and have developed methods to culture and expand these cells in the lab. We have also developed strategies for transplanting these cells into mice with Hirschsprung disease and other diseases that are caused by an absence or abnormality of the intestinal nervous system. Over the past few years, we have generated a variety of animal models for these studies and demonstrated that the transplanted cells are able to survive, grow, develop into neurons, and make functional connections with other cells after they are delivered into the recipient animal’s intestine. These studies confirm the feasibility of using cell therapy to treat these diseases and establish the foundation for human studies to be performed soon.
Enteric Nervous System Development
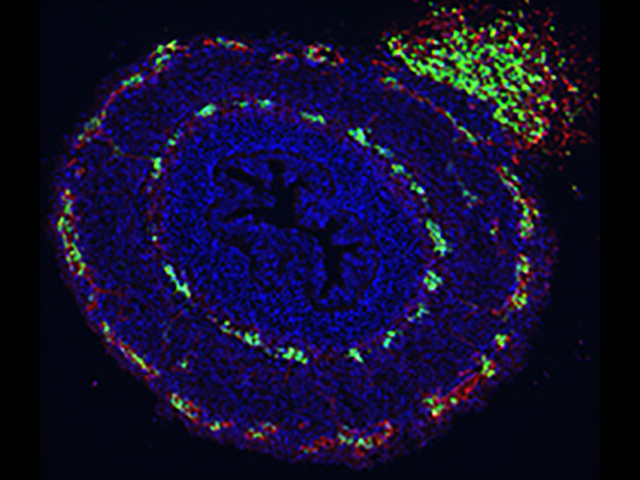
My lab is interested in how the enteric nervous system (ENS) develops in the embryo. The ENS is the complex neuronal network that exists in the gastrointestinal tract and whose principal function is to maintain normal intestinal motility. Congenital abnormalities of the ENS are responsible for severe gastrointestinal disorders in childhood, including Hirschsprung disease, intestinal pseudo-obstruction, esophageal achalasia, and other conditions that are caused by gut dysmotility. The ENS is made up of neurons and glia that arise from the neural crest and migrate along the length of the intestine.
Proteins in the intestinal microenvironment interact with factors expressed on the crest-derived cells to direct their migration, proliferation, survival, and differentiation. Hirschsprung disease results from the absence of ganglion cells along a variable length of distal intestine, leaving affected children with a severe functional obstruction. While several genetic mutations are associated with Hirschsprung disease, the mechanism of action of these genes remains poorly understood. Additionally, the genetic basis of other neurointestinal diseases is largely unknown.
We use the avian embryo as a model system to study ENS development. Using a variety of techniques, including retroviral-mediated gene misexpression, chick-quail chimeras, coelomic transplantation, and organ culture techniques, we have characterized normal development of the ENS and have identified important roles for endothelin-3, GDNF, extracellular matrix proteins, and Hedgehog signaling during ENS formation. Understanding the molecular factors required for normal ENS development will give us new insights into the pathophysiology of Hirschsprung disease and other neurointestinal disorders and allow us to develop novel diagnostic and therapeutic approaches to care for children with these serious conditions.
Clinical/Translational Research
Constipation is a common complaint in pediatrics, accounting for 25% of all visits to a pediatric gastroenterologist. While most children improve with diet modification or medications, some suffer from severe constipation that does not respond to conventional therapy. These children often have abdominal pain and bloating, fecal incontinence, absence from school, frequent hospitalizations, and social withdrawal. One of the major causes of this intractable constipation is abnormal colonic motility, where the colon is unable to propel waste products out of the body. This problem is seen in slow transit constipation, irritable bowel syndrome, Hirschsprung disease, and other disorders of colonic function. Many individuals of all ages are affected by these conditions. However, despite its prevalence, little is known about the causes of abnormal colonic motility and therefore available treatments are limited.
Children with Hirschsprung disease can suffer from severe constipation and bloating even after they have had surgery to remove the part of the colon lacking nerve cells. Their symptoms, which can be very difficult to treat, are often due to abnormal colonic motility or abnormal anorectal function. In some cases, children with Hirschsprung can develop enterocolitis, a life-threatening inflammation of the intestine. As in other conditions caused by abnormal intestinal motility, the causes are unknown and treatment options few.
Current research studies include:
- Examining the nerve cells, glial cells, and neurotransmitters in the intestine of children with Hirschsprung disease after they have surgery to understand the possible causes of persistent post-operative bowel problems.
- Using metabolite profiling to identify novel serum biomarkers associated with colonic contractility in children.
- Exploring minimally invasive approaches to visualize the enteric ganglia without the need for rectal or colonic biopsies.
- Identifying potential causes for Hirschsprung-associate enterocolitis using a genetic mouse model of the disease.

