Immunobiology Laboratory
Contact Information
Diabetes Unit Immunobiology Laboratory
Building 149
Room 3602
149 13th Street
Charlestown,
MA
02129
Phone: 617-726-4084
Fax: 617-643-9930
Email: diabetestrial@partners.org
Hours:
7:30 am-4:00 pm
Email Us at diabetestrial@partners.org
Phone: 617-726-4084
Explore This Research Lab
Overview
The Immunobiology Laboratory performs basic, translational and clinical research investigating new therapeutic concepts to treat immune disorders ranging from autoimmune disease to cancer. Under the direction of Denise Faustman, MD, PhD, the laboratory is currently conducting the BCG Human Clinical Trial Program to investigate the BCG vaccine as a treatment for established type 1 diabetes. Over 600 clinical trial subjects are under recruitment or already enrolled. Our current enrollment is focused on two different groups of children with type 1 diabetes, but we encourage type 1 diabetics of all ages to register with us at diabetestrial.org and you will receive updates.
Clinical Research
Our clinical research program is in type 1 diabetes. We are investigating whether repeat vaccination with a generic vaccine—called the bacillus Calmette-Guerin, or BCG, vaccine—can eliminate abnormal T cells, induce beneficial human regulatory T cells (Tregs) and reverse established type 1 diabetes in humans. Positive data from our Phase I study in humans were published in 2012. This data was repeated in two additional human studies in the last 5 years. A Phase II clinical trial is currently ongoing, as is a follow-up study to monitor the long-term effects of BCG vaccination in patients from the Phase I trial.
Most trials testing immune interventions in type 1 diabetes are conducted in new-onset diabetics. This clinical trial program is unique in focusing on patients with established, rather than newly diagnosed, type 1 diabetes. It is also unique in being a nonprofit drug development program (supported solely through philanthropic donations from the public and nonprofit organizations), as well as in seeking diabetes reversal using an inexpensive generic drug. For people with long standing type 1 diabetes the drug can restore blood sugars to near normal based on the ability of BCG to correct a defect in white blood cells; after BCG the type 1 diabetic white blood cells regulate blood sugars with high precision.
Basic Research
Our basic research program is focused on uncovering the basic molecular and immunological mechanisms behind human and murine immune pathogenesis and translating these findings into new innovations in the clinic. One of our findings has been that boosting or restoring tumor necrosis factor (TNF) can selectively eliminate pathogenic T cells, induce beneficial regulatory T cells (Tregs) and induce organ regeneration, permanently curing mice of both type 1 diabetes and Sjögren’s-like syndrome. This basic science research has led to the establishment of clinical trial programs using repeat BCG vaccination in diverse human autoimmune diseases, including the BCG Human Clinical Trial Program being conducted at Massachusetts General Hospital.
Other basic science research in our lab is focused on this discovery of novel tumor necrosis factor receptor superfamily receptor (TNFSFR) function and therapeutic use One of our fundamental basic science findings in the human is that tumor necrosis factor two (TNFR2) is an identifying surface protein of the most potent Tregs, and that the TNFR2 pathway is an important signaling pathway for adult Treg fate. Based on these findings, our lab has been making and testing unique antibodies for TNFR2 with the goal of identifying antibodies that can lead to human Treg proliferation and depletion, which may have future applications in immunotherapy for both autoimmunity and cancer. We also recently collaborated with a major pharmaceutical company to discover and develop a novel therapeutic antibody targeting another TNFSF receptor, CD40.
More information about the BCG human clinical trial program, please email diabetestrial@partners.org.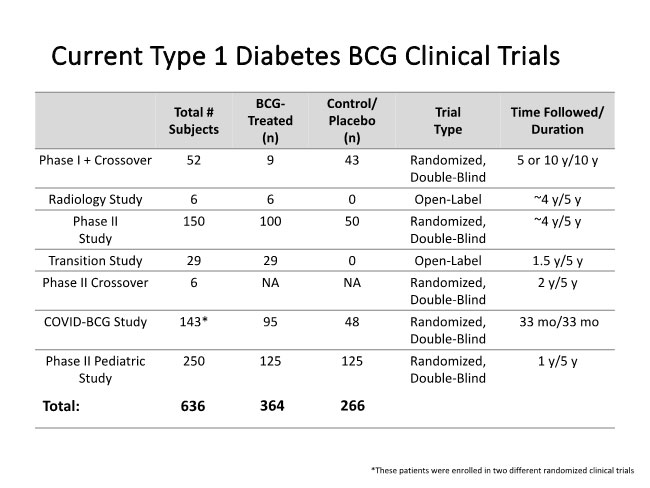
Faustman Lab
Visit the Faustman Lab website at faustmanlab.org.
Publications
The following publications were selected from over 200 peer reviewed articles.
- Faustman DL, Wang L, Okubo Y, Burger D, Ban L, Man G, Zheng H, Schoenfeld D, Pompei R, Avruch J, Nathan DM. Proof-of-concept, randomized, controlled clinical trial of Bacillus-Calmette-Guerin for treatment of long-term type I diabetes. PLoS ONE 2012; 7(8):e41756.
- Faustman DL, Davis M. TNF Receptor 2 and Disease: Autoimmunity and Regenerative Medicine. Front Immunol 2013; 4:478.
- Kuhtreiber WM, Leung SL, Wang L, Hsu E, Reinhold PE III, Zhao M, Zhang H, Burger DE, Faustman DL. Possible transient benefits of Epstein Barr virus infection in three subjects with established type 1 diabetes. J. Diabetes Metab 2013;4(9)309-314.
- Okubo Y, Mera T, Wang L, Faustman DL. Homogeneous expansion of human T-regulatory cells via tumor necrosis factor receptor 2. Nature Science Reports 2013; 3:3153. DOI 10.1038/srep03153
- Faustman DL. Why were we wrong for so long? The pancreas of type I diabetic patient commonly functions for decades. Diabetologia 2014;57(1):1-3. DOI 10.1007/s00125-013-3104-9.
- Mera T, Heimfeld S, Faustman DL. The spleen contributes stem cells to peripheral blood stem cell transplants. J Stem Cell Res Ther. 2014; 4:253.
- Faustman DL. Reversal of established autoimmune diabetes by in situ beta-cell regeneration. Ann NY Acad Sci 2002; 961:40. DOI: 10.1111/j.1749-6632.2002.tb03043.x.
- Faustman DL, Glesecke C, Davis M, Kuhtreiber WM, Tran SD, Dorners T, Ley EJ. Disposable no longer: The spleens holds a reservoir of stem cells. J Stem Cell Res Ther 2014; 4:7.
- Faustman DL, Rheinhold P, Washer SLL, Hsu E, Chao M, Burger D, Zheng H. Low levels of C-peptide production protect from complication and improve HbA1c, control in longstanding type 1 diabetes. Diabetologia 2014; 57:S45.
- Ban L, Kuhtreiber W, Butterworth J, Okubo Y, Vanamee ES, Faustman DL. Strategic internal covalent cross-linking of TNF produces a stable TNF trimer with improved TNFR2 signaling. Mol Cell Ther 2015; 3:7.
- Mera T, Faustman DL. Removal of donor human leukocyte antigen class I proteins with papain: translation for possible whole organ practices. Transplantation 2015; 99(4):724-730.
- Kuhtreiber WM, Washer SL, Hsu E, Zhao M, Rheinhold P 3rd, Burger D, Zheng H, Faustman DL. Low levels of C-peptide have clinical significance for established type I diabetes. Diabetic Med 2015; 32(10):1346-1353.
- Okubo Y, Torrey H, Butterworth J, Zheng H, Faustman DL. Treg activation defect in type I diabetes: correction with TNFR2 agonism. Clin Transl Immunology 2016; 5(1):e56. DOI 10.1038/cti.2015.43
- Torrey H, Butterworth J, Mera T, Okubo Y, Wang L, Baum D, Defusco A, Plager S, Warden S, Vanamee E, Foster R, Faustman DL. Targeting TNFR2 with antagonistic antibodies inhibits proliferation of ovarian cancer cells and tumor-associated Tregs. Sci Signal 2017; 10(482).
- Vanamee ES, Faustman DL. TNFR2: A novel target for cancer immunotherapy. Trends Mol Med 2017; 23(11):1037-1046.
- Kuhtreiber WM, Tran L, Kim T, Dybala M, Nguyen B, Plager S, Huang D, Janes S, Defusco A, Baum D, Zheng H, Faustman DL. Long-term reduction in hyperglycemia in advanced type I diabetes: the value of induced aerobic glycolysis with BCG vaccinations. Nature Vaccines 2018; 3:23.
- Faustman DL. TNF, TNF inducers, and TNFR2 agonists: A new path to type I diabetes treatment. Diabetes Metab Res Rev 2018;34(1)
- Vanamee ES, Faustman DL. Structural principles of tumor necrosis factor superfamily signaling. Science Signaling 2018; 11(511)
- Atretkhany KN, Mufazalov IA, Dunst J, Kuchmly A, Gogoleva VS, Andruszewski D, Drutskaya MS, Faustman DL, Schwabenland M, Prinz M, Kruglov AA, Waisman A, Nedospasov SA. Intrinisic TNFR2 signaling in T regulatory cells provides protection in CNS autoimmunity. PNAS 2018; 115(51):13051-13056.
- Ristori G, Faustman D, Matarese G, Romano S, Salvetti M. Bridging the gap between vaccination with Bacille Calmette-Guerin (BCG) and immunological tolerance: the cases of type I diabetes and multiple sclerosis. Curr Opin Immunol 2018; 55:89-96.
- Kuhtreiber WM, Faustman DL. BCG therapy for type 1 diabetes: restoration of balance immunity and metabolism. Cell: Trends Endocrinol Metab 2019; 30(2):80-92.
- Torrey H, Kuhtreiber WM, Okubo Y, Tran L, Case K, Zheng H, Vanamee E, Faustman DL. A novel TNFR2 agonist antibody expands highly potent regulatory T cells. Sci Signal 2020; 13(661) eaba9600. DOI 10.1126/scisignal.aba9600.
- Torrey H, Khodadoust M, Tran L, Baum D, Defusco A, Kim YH, Faustman DL. Targeted killing of TNFR2 expressing tumor cells and Tregs by TNFR2 antagonistic antibodies in advanced Sezary syndrome. Nature Leukemia 2019; 33(5):1206-1218.
- Kuhtreiber WM, Faustman DL. BCG therapy for type I diabetes: restoration of balance immunity and metabolism. Trends Endocrinol Metab 2019; 30(2):80-92. DOI 10.1016/j.tem.2018.11.006.
- Yang M, Tran L, Torrey H, Song Y, Perkins H, Case K, Zheng H, Takahashi H, Kuhtreiber WM, Faustman DL. Optimizing TNFR2 antagonism for immunotherapy with tumor microenvironment specificity. J Leukoc Biol 2020; 107(6):971-980.
- Case K, Tran L, Yang M, Zheng H, Kuhtreiber WM, Faustman DL. TNFR2 blockage alone or in combination with PD-1 blockade shows therapeutic efficacy in murine cancer modes. J Leukocye Biol 2020; 107(6):981-991. DOI 10.1002/JLB.5MA0420-375RRRRR.
- Kuhtreiber WM, Takahashi H, Keefe RC, Song Y, Tran L, Luck TG, Shpilsky G, Moore L, Sinton SM, Graham JC, Faustman DL. BCG vaccinations upregulate Myc, a central switch for improved glucose metabolism in diabetes. Cell iScience 2020; 23(5):101085.
- Faustman DL, Kuhtreiber W. Benefits of BCG induced metabolic switch from oxidative phosphorylation to aerobic glycolysis in autoimmune and nervous system diseases. J Intern Med 2020; 288:641-650.
- Vanamee E, Faustman DL. On the TRAIL of better therapies: Understanding TNFRSF structure-function Cell 2020; 9:764.
- Vanamee ES, Faustman DL. On the TRAIL of better therapies: Understanding TNFRSF structure-function. Cells 2020; 9(3) 764. DOI 10.3390/cells9030764.
- Faustman DL. Benefits of BCG-induced metabolic switch from oxidative phosphorylation to aerobic glycolysis to aerobic glycolysis in autoimmune and nervous system diseases. J Intern Med 2020; 288(6):641-650.
- Dias HF, Kuhtreiber WM, Nelson KJ, Ng NC, Zheng H, Faustman DL. Epigenetic changes related to glucose metabolism in type 1 diabetes after BCG vaccinations. A vital role for KDM2B. Nature Vaccines, 2022;40:1540-1554. DOI 10.1016/j.vaccine.2021.04.011. doi:10.1016/j.vaccine.2021.04.011.
- Rivero-Calle I, Gomez-Rial J, Bont L, Gessner BD, Kohn M, Dagan R, Payne DC, Bruni L, Pollard AJ, Garcia-Sastre A, Faustman DL, Osterhaus A, Butler R, Gimenez Sanchez F, Alvarez F, Kaforou M, Bello X, Martinon-Torres F. TIPICO X: A report of the 10th interactive infectious disease workshop on infectious diseases and vaccines. Human Vaccine Immunother 2021; 17(3):759-772.
- Keefe, R, Takahshi, H, Tran, L, Nelson, K, Ng, N, Kuhtreber, W, Faustman, DL. BCG therapy is associated with long term durable induction of Treg signature genes by epigenetic modulation. Nature Scientific Reports 2021;11:14933-14945.
- Shpilsky GF, Takahashi H, Aristarkhova A, Weil M, Ng N, Nelson KJ, Lee A, Zheng H, Kühtreiber WM, Faustman DL. Bacillus Calmette-Guerin 's beneficial impact on glucose metabolism: evidence for broad based applications. iScience. 2021 Sep 21;24(10):103150.
- Vanamee ÉS, Lippner G, Faustman DL. Signal Amplification in Highly Ordered Networks Is Driven by Geometry. Cells. 2022 Jan 13;11(2):272
- Takahashi H, Yoshimatsu G, Faustman DL. The Roles of TNFR2 Signaling in Cancer Cells and the Tumor Microenvironment and the Potency of TNFR2 Targeted Therapy. Cells. 2022 Jun 17;11(12):1952.
- Faustman DL, Lee A, Hostetter ER, Aristarkhova A, Ng NC, Shpilsky GF, Tran L, Wolfe G, Takahashi H, Dias HF, Braley J, Zheng H, Schoenfeld DA, Kühtreiber WM. Multiple BCG vaccinations for the prevention of COVID-19 and other infectious diseases in type 1 diabetes. Cell Rep Med. 2022 Sep 20;3(9):100728.
- Takahashi H, Kühtreiber WM, Keefe RC, Lee AH, Aristarkhova A, Dias HF, Ng N, Nelson KJ, Bien S, Scheffey D, Faustman DL. BCG vaccinations drive epigenetic changes to the human T cell receptor: Restored expression in type 1 diabetes. Sci Adv. 2022 Nov 18;8(46):eabq7240.
- Dias HF, Mochizuki Y, Kühtreiber WM, Takahashi H, Zheng H, Faustman DL. Bacille Calmette Guerin (BCG) and prevention of types 1 and 2 diabetes: Results of two observational studies. PLoS One. 2023 Jan 20;18(1):e0276423.
- Vanamee ÉS, Faustman DL. The benefits of clustering in TNF receptor superfamily signaling. Front Immunol. 2023 Aug 17;14:1225704.
