Bacskai Lab at MIND


Contact Information
MassGeneral Institute for Neurodegenerative Disease
Building 114, Charlestown Navy Yard
114 16th Street, Room 2003
Mailcode: CNY B114-2-2003
Charlestown,
MA
02129
Phone: 617-726-2000
Fax: 617-724-1480
Fundraising Contact
Contact Krista McCabe in the Mass General Development office at 617-726-1392 or kmccabe@partners.org.
Please visit the main Neurology Service Contact page for all other contact information.
Publications
Explore This Lab
Overview
Using the mulitphoton micrsocopy imaging technique, senile plaques of Alzheimer's disease can be detected in the brains of living transgenic mice, and characterized with chronic imaging.
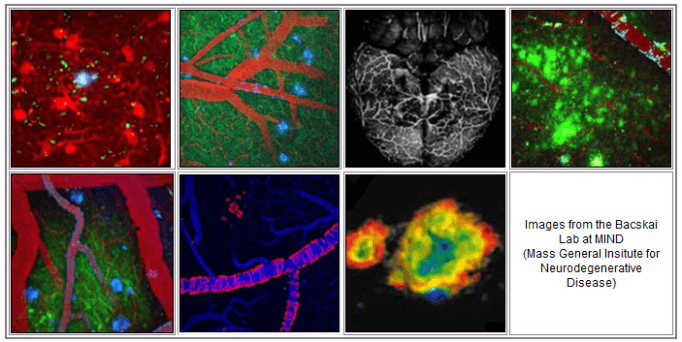
This detection platform was used to characterize a therapeutic approach to clearing the senile plaques based on immunotherapy, as well as to characterize novel amyloid-targeting ligands in preclinical development for PET imaging in humans.
Current research is aimed at optimizing anti-amyloid- therapeutic approaches, and imaging the anatomy and physiology of specific cell types in the brain before and after treatment. Development of novel optical techniques is ongoing, and includes methods to measure protein-protein interactions using fluorescence lifetime imaging microscopy (FLIM), and non-invasive approaches to amyloid imaging in intact animals.
Research Projects
Multiphoton microscopy
We use multiphoton microscopy to image the brains of living mice at very high resolution. The technique relies on non-linear excitation of fluorophores with a femto-second pulsed near infrared laser. This allows deep tissue imaging with reduced photo-toxicity, making it ideal for penetrating hundreds of microns into brain. The approach excites multiple fluorophores simultaneously for multiplexing experiments and can be used to detect targeted cells and structures for longitudinal imaging over periods of days, weeks, months, or even a year.
Brain imaging in mouse models of disease
Alzheimer’s disease is a strictly human disease, so models are required to identify the cellular and molecular factors involved in the progression of the disease. Transgenic mouse models are powerful, in that they are engineered to develop the pathologies found in human brain tissue. This allows us to dissect the specific effects of the progression of plaques and tangles on the cellular and molecular components of the intact brain.
Neuronal structure and function
Neurons are obviously the main target of "neurodegenerative diseases" and can be easily imaged with structural and functional markers. We use fluorescent protein constructs to measure neurite morphology and spine loss, and functional markers that report on static and dynamic calcium concentration and redox potential. These probes can be targeted to specific compartments within a neuron including nucleus, mitochondria, and endoplasmic reticulum.
Astrocyte structure and function
Astrocytes have long been considered a “support” cell in the brain, and occupy more than half of brain volume. It is increasingly clear that they play an active role in signaling, synapse maintenance, and direct interactions with the cerebrovasculature (neurovascular coupling). Additionally, as a cellular component of neuroinflammation, astrocytes respond to damage (e.g. deposition of senile plaques) likely playing a protective role. This “gliosis” is a well-established feature of Alzheimer’s disease, but is poorly understood.
Cerebrovascular contributions to disease
What is often overlooked in reduced preparations for studying the brain, like cell cultures and acute slices, is the role of the cerebrovasculature. Alterations in blood flow are a primary component of Alzheimer’s disease, and on their own can lead to cognitive deficits. Using intact, living brains, we can image specific cell types in the brain along with the blood vessels (both structure and function). In this way, we can image the neurovascular unit – neurons, astrocytes, and blood vessels – during resting or evoked responses, and evaluate the potential alterations that occur during senile plaque and cerebral amyloid angiopathy (CAA) deposition in the mouse models.
Non-invasive NIR imaging
Multiphoton microscopy is a powerful approach that allows sub-micron resolution several hundred microns deep into the brain. However, the technique relies on a surgical approach to remove skin and skull. Ideally, it should be possible to use non-invasive imaging with near infrared excitation and emission to image the whole brain, albeit at much reduced spatial resolution. While the techniques have been developed, the field lacks suitable fluorophores in this spectral range that engage with specific targets in the brain. We have successfully identified candidate fluorophores that label senile plaques, that ultimately should allow us to measure the amyloid load in the transgenic mice without surgical intervention. Additionally, this approach could allow non-invasive detection in humans, at much lower cost than PET or SPECT imaging and without the need for radioactive tracers.
Group Members
Brian J. Bacskai, PhD
- Professor of Neurology, Harvard Medical School
- Principal Investigator, Neurology, Massachusetts General Hospital
Lab Members
- Maria Calvo Rodriguez, PhD
- Patricia Kelly, PhD
- Rebecca Gillani, MD, PhD
- Steve Hou, PhD
- Susanne Van Veluw, PhD
Research Positions
Read about and apply for residency, fellowship and observership programs in pathology or in neurology.
All applicants should register with the Mass General Careers website.
Request a list of current open Neurology positions at mghneurologyjobs@partners.org.
Support MIND
Your contributions help drive discovery and propel findings from the laboratory bench into the clinic.
