MUCOSAL IMMUNOLOGY & BIOLOGY RESEARCH
Faherty Lab: Christina Faherty, PhD
Contact Information
Faherty Lab
Building 114, 16th Street
Charlestown,
MA
02129
Phone: 617-726-4166
Email: csfaherty@partners.org
Explore This Lab
Overview
The focus of the Laboratory of Christina Faherty, PhD, at Massachusetts General Hospital is enhancing our understanding of gastrointestinal bacterial pathogens, particularly the Shigella species.
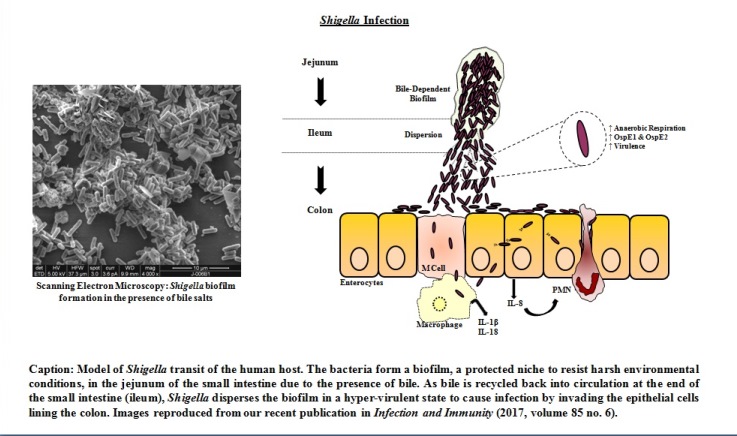
Shigella causes a significant global health burden each year by causing millions of infections predominantly in children under the age of five years in developing countries. Children subjected to recurrent diarrhea in developing countries often exhibit environmental enteropathy, a condition defined by severe damage to the gastrointestinal tract that typically results in delayed growth and development. There is still no effective vaccine against Shigella, antibiotic resistance is complicating disease treatment, and foodborne infections in industrialized nations are on the rise.
While many aspects of Shigella colonic cell invasion and intracellular survival are known, crucial gaps in knowledge remain that have hindered successful vaccine development. To cause infection in the colon, Shigella must transit the majority of the gastrointestinal tract and survive detrimental conditions such as a low pH in the stomach and bile in the small intestine. We seek to understand how Shigella alters gene expression during host transit and to determine how the bacteria recognize the colonic environment to initiate infection. By focusing on these processes, we will understand the early events in Shigella infection and identify unique virulence factors to target for vaccine development. Our work has enabled us to expand the Shigella infection paradigm and demonstrate the importance of utilizing in vivo stimuli to mimic host transit in our research.
In all, we have a better understanding of Shigella pathogenesis that could finally translate to successful vaccine candidates and improve the health of millions of people around the globe.
Research Projects
Identify the adherence factors utilized by Shigella to initiate contact with the colonic epithelium
Our previous work demonstrates that S. flexneri exposure to bile salts induces the expression and outer membrane localization of OspE1 and OspE2, two proteins that function as adhesins to initiate host cell contact.
We are currently expanding our work with OspE1 and OspE2 as well as determining if other adherence factors are utilized by the bacteria.
This work uses novel models of the human gastrointestinal tract to facilitate characterization and discovery of adherence factors.
Understand the gene expression profile during host transit
We are currently utilizing various host signals to understand Shigella survival and how virulence factors are expressed during gastrointestinal transit.
Given the efficient invasion mechanism of Shigella, we hypothesize that host factors serve as important signals to optimize virulence gene expression at key points prior to colonic epithelial cell contact.
We are also expanding our analyses to clinical isolates of Shigella to compare the various Shigella species, investigate if differences in virulence explain epidemiological infection patterns around the globe and determine if a multivalent vaccine can be developed.
With collaborators at Massachusetts Institute of Technology (MIT), we are designing and engineering bacteriophage viruses that will target and kill Shigella and other enteric pathogens while leaving friendly commensal bacteria unharmed.
It is our hope that phage therapy will become a viable alternative to antibiotics and enhance treatment for environmental enteropathy.
News & Events
- Novel models for enteric infections—an interview with the Faherty Lab
- Bile salt-induced biofilm formation in enteric pathogens: techniques for identification and quantification
- Harmful bacteria survive perilous path through the gut with help from an unlikely source
- CDC notes Shigella strains with reduced susceptibility
- Shigella investigators in Michigan bring in help from the CDC
- Shigellosis outbreak at elementary school kept quiet for weeks
- Multidrug-resistant Shgellosis: An Emerging Sexually Transmitted Infection
- Outbreak sickens 167 in Dubuque County, Iowa
- Galveston county officials mark rise in gastrointestinal illness
Lab Members

Principal Investigator
Christina S. Faherty, PhD
Assistant Molecular Biologist, Massachusetts General Hospital
Assistant Professor of Pediatrics, Harvard Medical School
Research Team
Meryl Perlman, MD, Pediatric Gastroenterology FellowJames Carey, Masters thesis student at Boston University
Alumni
Kourtney Nickerson, PhD: Charles River Laboratories
Rachael B. Chanin: Currently enrolled in a PhD program at the University of Texas Southwestern Medical Center
Alejandro Llanos-Chea, MD: Assistant Professor of Pediatrics at the University of Miami, Miller School of Medicine
Publications
Selected Publications
Szabady RL, Louissaint C, Lubben A, Xie B, Reeksting S, Tuohy C, Demma Z, Foley SE, Faherty CS, Llanos-Chea A, Olive AJ, Mrsny RJ, McCormick BA. Intestinal P-glycoprotein exports endocannabinoids to prevent inflammation and maintain homeostasis. J Clin Invest. 2018 Aug 31;128(9):4044-4056. doi: 10.1172/JCI96817. Epub 2018 Aug 13. PubMed PMID: 30102254
Llanos-Chea A, Citorik RJ, Nickerson KP, Ingano L, Serena G, Senger S, Lu TK, Fasano A, Faherty CS. Bacteriophage Therapy Testing Against Shigella flexneri in a Novel Human Intestinal Organoid-Derived Infection Model. J Pediatr Gastroenterol Nutr. 2019 Apr;68(4):509-516. doi: 10.1097/MPG.0000000000002203. PubMed PMID: 30418409.
Nickerson KP and Faherty CS. Bile Salt-induced Biofilm Formation in Enteric Pathogens: Techniques for Identification and Quantification. J Vis Exp. 2018 May 6;(135). doi: 10.3791/57322. PMID: 29781989.
Nickerson KP, Senger S, Zhang Y, Lima R, Patel S, Ingano L, Flavahan WA, Kumar DKV, Fraser CM, Faherty CS, Sztein MB, Fiorentino M, Fasano A. Salmonella Typhi Colonization Provokes Extensive Transcriptional Changes Aimed at Evading Host Mucosal Immune Defense During Early Infection of Human Intestinal Tissue. EBioMedicine. 2018 May;31:92-109. doi: 10.1016/j.ebiom.2018.04.005. PMID: 29735417.
Nickerson KP, Chanin RB, Sistrunk JR, Rasko DA, Fink PJ, Barry EM, Nataro JP, Faherty CS. Analysis of Shigella flexneri Resistance, Biofilm Formation, and Transcriptional Profile in Response to Bile Salts. Infect Immun. 2017 May 23;85(6). pii: e01067-16. PMID: 28348056
Faherty CS, Wu T, Morris CR, Grassel CL, Rasko DA, Harper JM, Shea-Donohue T, Fasano A, Barry EM. The Synthesis of OspD3 (ShET2) in Shigella flexneri is independent of OspC1. Gut Microbes. 2016 Nov; 7(6):486-502. PMID: 27657187.
Sistrunk JR, Nickerson KP, Chanin RB, Rasko DA, Faherty CS. Survival of the Fittest: How Bacterial Pathogens Utilize Bile To Enhance Infection. Clin Microbiol Rev. 2016 Oct; 29(4):819-36. PMID: 27464994.
Faherty CS, Harper JM, Shea-Donohue T, Barry EM, Kaper JB, Fasano A, Nataro JP. Chromosomal and plasmid-encoded factors of Shigella flexneri induce secretogenic activity ex vivo. PLoS One. 2012; 7(11):e49980. PMID: 23166804; PMCID: PMC3500342.
Faherty CS, Redman JC, Rasko DA, Barry EM, Nataro JP. Shigella flexneri effectors OspE1 and OspE2 mediate induced adherence to the colonic epithelium following bile salts exposure. Mol Microbiol. 2012 Jul; 85(1):107-21. PMID: 22571618; PMCID: PMC3399131.
Ruiz-Perez F, Wahid R, Faherty CS, Kolappaswamy K, Rodriguez L, Santiago A, Murphy E, Cross A, Sztein MB, Nataro JP. Serine protease autotransporters from Shigella flexneri and pathogenic Escherichia coli target a broad range of leukocyte glycoproteins. Proc Natl Acad Sci U S A. 2011 Aug 2; 108(31):12881-6. PMID: 21768350; PMCID: PMC3150873.
Faherty CS, Merrell DS, Semino-Mora C, Dubois A, Ramaswamy AV, Maurelli AT. Microarray analysis of Shigella flexneri-infected epithelial cells identifies host factors important for apoptosis inhibition. BMC Genomics. 2010; 11:272. PMID: 20429941; PMCID: PMC2996966.
Faherty CS, Maurelli AT. Spa15 of Shigella flexneri is secreted through the type III secretion system and prevents staurosporine-induced apoptosis. Infect Immun. 2009 Dec; 77(12):5281-90. PMID: 19805534; PMCID: PMC2786474.
Zurawski DV, Mumy KL, Faherty CS, McCormick BA, Maurelli AT. Shigella flexneri type III secretion system effectors OspB and OspF target the nucleus to downregulate the host inflammatory response via interactions with retinoblastoma protein. Mol Microbiol. 2009 Jan; 71(2):350-68. PMID: 19017275; PMCID: PMC2783611.
Faherty CS, Maurelli AT. Staying alive: bacterial inhibition of apoptosis during infection. Trends Microbiol. 2008 Apr; 16(4):173-80. PMID: 18353648; PMCID: PMC2746948.
Clark CS*, Maurelli AT. Shigella flexneri inhibits staurosporine-induced apoptosis in epithelial cells. Infect Immun. 2007 May; 75(5):2531-9. PMID: 17339354; PMCID: PMC1865761. *Maiden Name
