We are committed to supporting the rapidly growing clinical trials portfolio with the goal of biologically optimizing radiation therapy.
Integrating Radiation Therapy with Immunotherapy
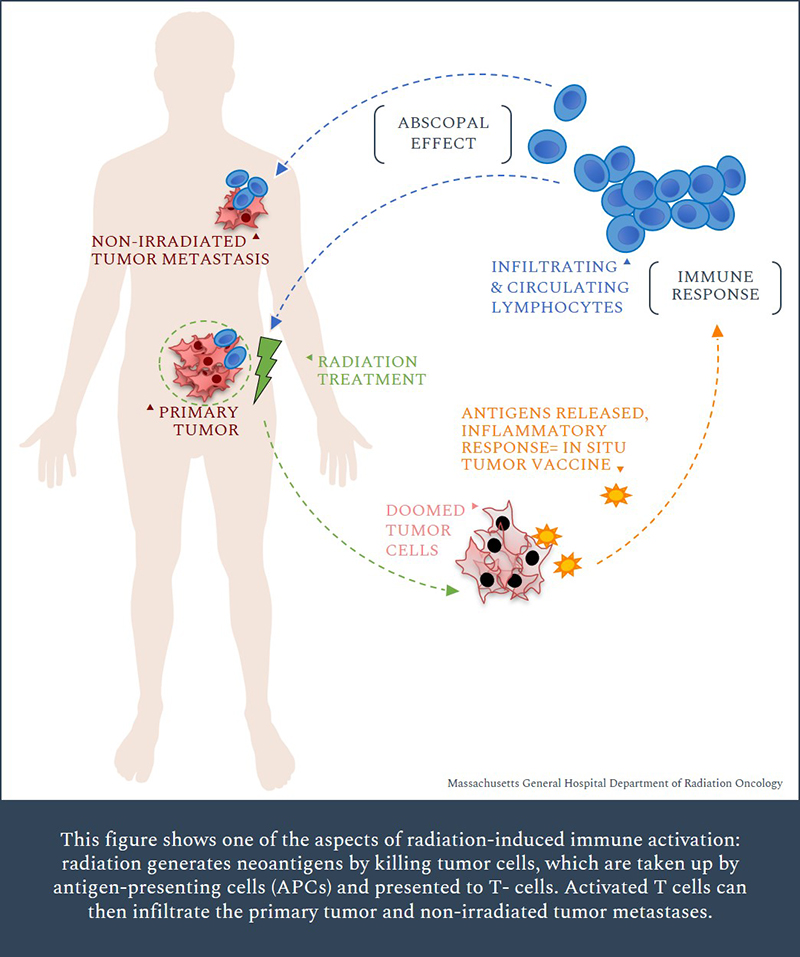 Immunotherapy is altering the landscape of cancer treatment, using the immune system to attack and destroy cancer cells. Combined with radiation therapy, the two therapies can work synergistically with the immune system by turning the tumor into a localized vaccine. A wealth of data has shown the interactions between radiation and the immune system, and it is now widely accepted that the therapeutic effect of ionizing radiation contains an immune-mediated component, necessitating the presence of immune cells.
Immunotherapy is altering the landscape of cancer treatment, using the immune system to attack and destroy cancer cells. Combined with radiation therapy, the two therapies can work synergistically with the immune system by turning the tumor into a localized vaccine. A wealth of data has shown the interactions between radiation and the immune system, and it is now widely accepted that the therapeutic effect of ionizing radiation contains an immune-mediated component, necessitating the presence of immune cells.
The effects of radiotherapy encompass a spectrum of mechanisms, which also include the destruction of tumor-associated stroma, the promotion of signals that guide the transition from non-specific to adaptive immune responses, and other factors. This increasingly complex landscape of therapy choices necessitates models that connect the quantitative response assessments of the tumor, its subpopulations, and the immune system to clinical decision-making.
Our researchers rely on information about our patients and their specific cancers to inform this decision-making; your participation in clinical trials will help guide our data-driven approach.
Radiation Therapy Combined with Molecular Targeted Agents
Radiation can be combined with chemotherapy and targeted agents to enhance the eradication of visible tumors to improve survival outcomes. Targeted agents are selected according to the genomic characteristics of a cancer, which requires additional testing.
The current practice of radiation oncology is primarily based on precise technical delivery of highly conformal, image-guided radiation treatments. The precision medicine revolution has provided radiation oncologists with tremendous opportunities to enhance the anti-tumor effects of radiation therapy, potentially with less normal tissue toxicity than traditional chemotherapeutic radiosensitizers. However, a large body of pre-clinical research and clinical investigations on molecular targeted radiosensitizers has not yet translated into any meaningful number of FDA-approved combinations of radiation with these agents. There exist distinct challenges to clinical translation of radiation/drug combinations that the field is only beginning to appreciate.
To address this, Mass General has launched an NCI-funded translational research initiative to accelerate the pace by which promising combinations of radiation, chemotherapy, and targeted radio-sensitizing agents are identified and subsequently clinically tested. Our researchers work closely with the Cancer Center to investigate tumor models and a panel of agents to accelerate advances in this novel field.



 Immunotherapy is altering the landscape of cancer treatment, using the immune system to attack and destroy cancer cells. Combined with radiation therapy, the two therapies can work synergistically with the immune system by turning the tumor into a localized vaccine. A wealth of data has shown the interactions between radiation and the immune system, and it is now widely accepted that the therapeutic effect of ionizing radiation contains an immune-mediated component, necessitating the presence of immune cells.
Immunotherapy is altering the landscape of cancer treatment, using the immune system to attack and destroy cancer cells. Combined with radiation therapy, the two therapies can work synergistically with the immune system by turning the tumor into a localized vaccine. A wealth of data has shown the interactions between radiation and the immune system, and it is now widely accepted that the therapeutic effect of ionizing radiation contains an immune-mediated component, necessitating the presence of immune cells.