Krantz Family Center for Cancer Research
McClatchey Lab


Contact Information
Andrea I. McClatchey, PhD
Director of Academic Affairs
Krantz Family Center for Cancer Research
Professor of Pathology
Harvard Medical School
Poitras Family Endowed Chair in Oncology
Program Affiliations
Krantz Family Center for Cancer Research
Massachusetts General Hospital Pathology Service
Explore the McClatchey Lab
Research Summary
The McClatchey laboratory focuses on understanding how cells spatially organize their outer surface to build and regenerate functioning tissues and how defects in that organization can drive tumor formation. Using in vivo and physiologic 3D models and quantitative imaging, we have uncovered important ways cells spatially coordinate receptor tyrosine kinase trafficking and signaling with cell-cell adhesion and actomyosin organization during tissue morphogenesis. Our studies have focused on the process of de novo lumen formation that drives morphogenesis of tubular bile ducts and other organs, and on the polarization and plasticity of Schwann cells in the peripheral nervous system. In each case, we uncovered unexpected ways that tumor-causing mutations hijack the mechanisms that normally spatially pattern the cell surface to drive tumor initiation and heterogeneity. Our research establishes new models and therapeutic avenues for both biliary (cholangiocarcinoma) and Schwann cell (schwannoma) tumors and highlights the value of convergent studies of morphogenesis and tumorigenesis.
Research Projects
Convergent studies of morphogenesis and tumorigenesis
The vast array of forms and functions exhibited by different cell types is enabled by the intrinsic organization of specialized domains within the cell cortex such as the leading edge of migratory cells, immunological synapse, and microvillusstudded apical surfaces of epithelial cells. The spatial organization of individual cells, in turn, governs their organization into three-dimensional structures that carry out organ-specific functions, such as the tubular networks of the liver, kidney, breast and lung and the heterotypic axoglial junction of peripheral nerves. The spatial organization of cortical domains in individual cells and tissues provides an essential layer of regulation to both biochemical and adhesive receptors on the cell surface. Alterations in cellular architecture are the earliest evidence of a developing tumor and signatures of tumor invasion and metastasis.
The overarching goal of my laboratory is to understand how the dynamic organization of the outer cell surface contributes to morphogenesis and tumorigenesis. We have focused particular attention on the liver and peripheral nervous system. For example, we discovered that biliary epithelial cells self-organize into a tubular network during development via the de novo formation, extension and interconnection of apical lumens. Using a physiologic and quantitative 3D model we found that FGFR signaling is important for biliary morphogenesis, and that FGFR2 mutants that are common drivers of cholangiocarcinoma disrupt this process. Unexpectedly, we found that the trafficking and signaling of normal FGFR2 and phenotypic consequences of FGFR2 mutants are governed by the epithelial state of the cell and nature of the mutation, highlighting the value of using mutants as tools to study morphogenesis and of physiological models to study tumor-causing mutants.
Related studies center on a longstanding focus on the role of the membrane:cytoskeleton linking neurofibromatosis type 2 (NF2) tumor suppressor, Merlin, and closely related ERM proteins (Ezrin, Radixin and Moesin) in organizing the Schwann cell surface, We found that in the absence of Merlin, Schwann cells exhibit unstable polarity, which, in turn, yields intrinsic heterogeneity in an otherwise genetically ‘cold’ (homogeneous) tumor. Through quantitative imaging in mouse models we created an initial atlas of how schwannoma heterogeneity develops, evolves and responds to drug treatment. These studies have provided desperately needed insight into the histological, clinical and therapeutic heterogeneity exhibited by schwannomas and a framework for overcoming it – something desperately needed for NF2 patients who often develop multiple debilitating spinal and cranial schwannomas.
Publications
View a list of publications by researchers at the McClatchey Laboratory
Selected Publications
O’Loughlin E, Zhang Y, ChiassonMacKenzie C, Dave P, Rheinbay E, Stott S, McClatchey AI. Distinct phenotypic consequences of cholangiocarcinoma-associated FGFR2 alterations depend on epithelial maturity. bioRxiv, 10.1101/2024.08.30.610360, 2024.
Chiasson-MacKenzie C, Vitte J, Liu CH, Wright EA, Flynn EA, Stott SL, Giovannini M and McClatchey AI. Cellular mechanisms of heterogeneity in NF2-mutant schwannoma. Nat Comm 14(1):1559, 2023 March 21.
Chiasson-MacKenzie C, Morris ZS, Liu CH, Bradford WB, Koorman T, McClatchey AI. Merlin/ERM proteins regulate growth factor-induced macropinocytosis and receptor recycling by organizing the plasma membrane:cytoskeleton interface. Genes Dev. 32(17-18): 1201-14, 2018 Sep 1.
Benhamouche-Trouillet S, O’Loughlin E, Liu CH, Polachek W, Fitamant J, McKee M, El-Bardeesy N, Chen CS, McClatchey AI. Proliferation-independent role of NF2/Merlin in limiting biliary morpho- genesis. Development 145(9), 2018 April 30.
Chiasson-MacKenzie C, Morris ZS, Baca Q, Morris B, Coker JK, Mirchev R, Jensen AE, Carey T, Stott SL, Golan DE, McClatchey AI. NF2/Merlin mediates contact-dependent inhibition of EGFR mobility and internalization via cortical actomyosin. J Cell Biol. 2015 Oct 26;211(2):391-405.
Hebert AM, DuBoff B, Casaletto JB, Gladden AB, McClatchey AI. Merlin/ERM proteins establish cortical asymmetry and centrosome position. Genes Dev. 2012 Dec 15;26(24):2709-23.
Benhamouche S, Curto M, Saotome I, Gladden AB, Liu CH, Giovannini M, McClatchey AI. Nf2/Merlin controls progenitor homeostasis and tumorigenesis in the liver. Genes Dev. 2010 Aug 15;24(16):1718-30.
*denotes equal contribution
Research Image
Left: Digital image analysis highlights intra-tumoral heterogeneity of autocrine ligand production in a dorsal root ganglia from a six-month old Postn-Cre/Nf2flox/flox mouse. The Highplex FL algorithm in HALO imaging software was used to achieve single cell segmentation and detect neuregulin-1 positive (magenta), phospho-S6 positive (green), or neuregulin-1/phospho-S6 positive (gray) cells (in collaboration with the lab of Dr. Stott). Image: Christine-Chiasson MacKenzie, PhD
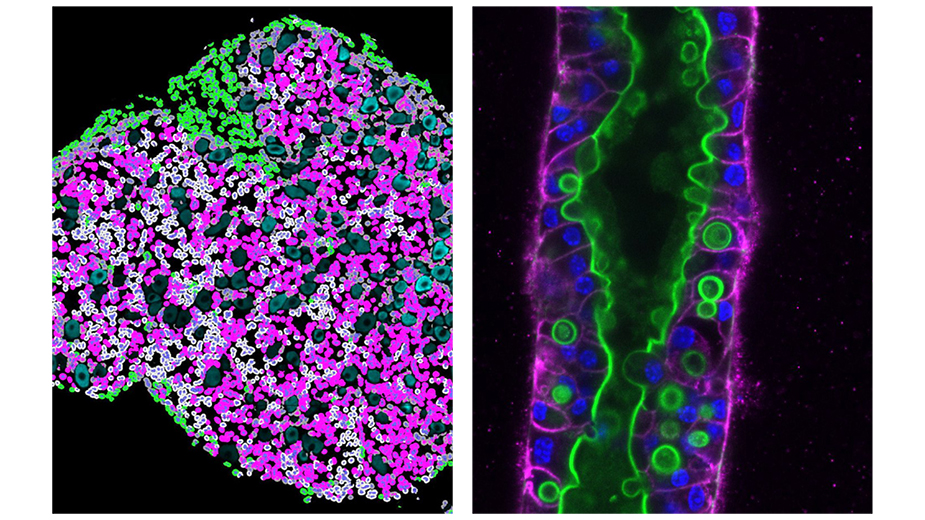
Right: Confocal image of a three dimensional cell culture model of biliary tube formation labelled for E-cadherin (green) and actin (magenta). Image credit: Evan O’Loughlin, PhD
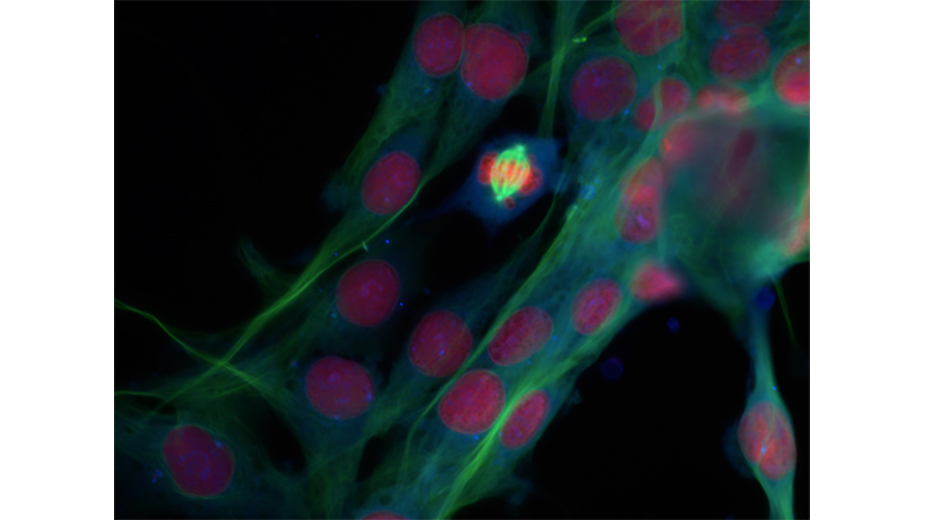
Image: McClatchey lab

Image: McClatchey lab

Image: McClatchey lab
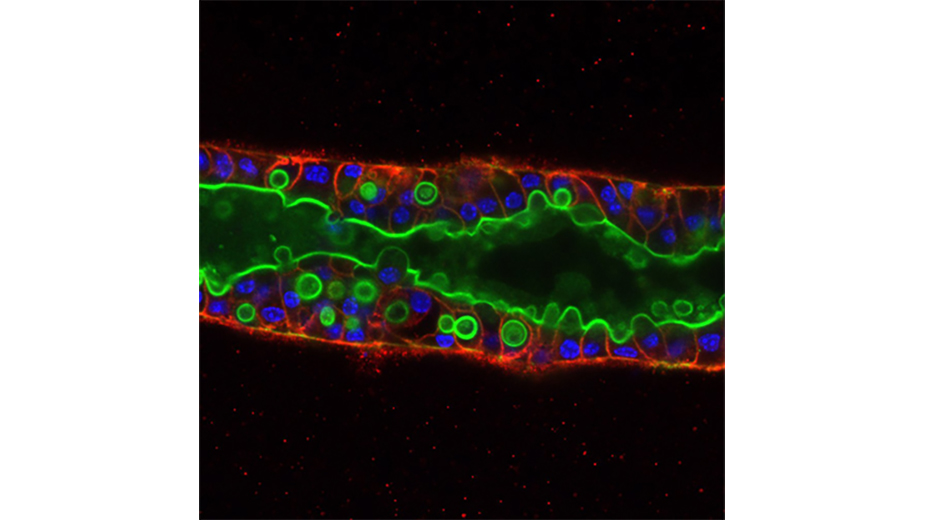
Image: McClatchey lab
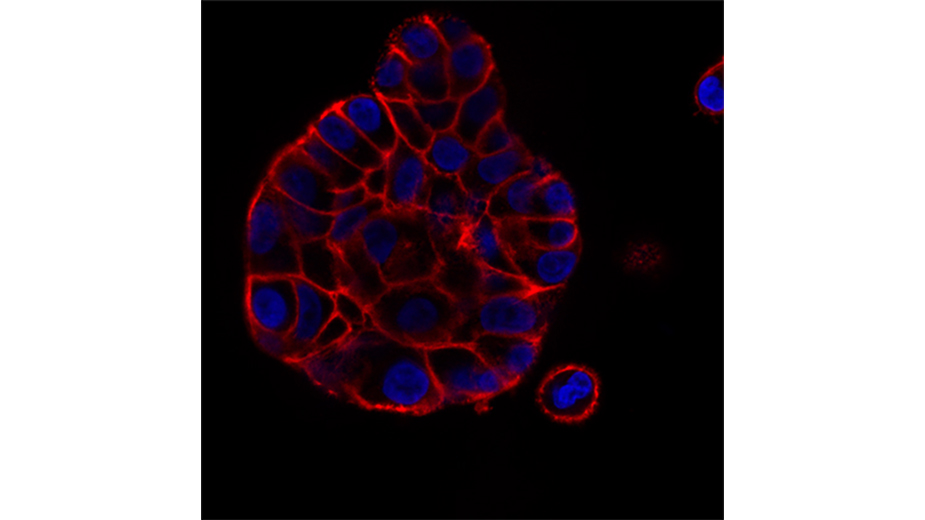
Image: McClatchey lab
Our Researchers
Andrea I. McClatchey, PhD
Group Members
- Sarah Bushnell
- Christine Chiasson-MacKenzie, PhD
- Catherine Movsessian
- Ashley Westerfield, PhD
- Emily A. Wright
Krantz Family Center for Cancer Research
The scientific engine for discovery for the Mass General Brigham Cancer Institute.
Support the Krantz Family Center for Cancer Research
When you support us you are enabling discoveries that will lead to effective new weapons in the battle against cancer.
