Department of Surgery
Center for Engineering in Medicine & Surgery
About the CEMS
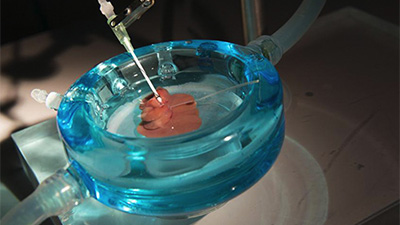
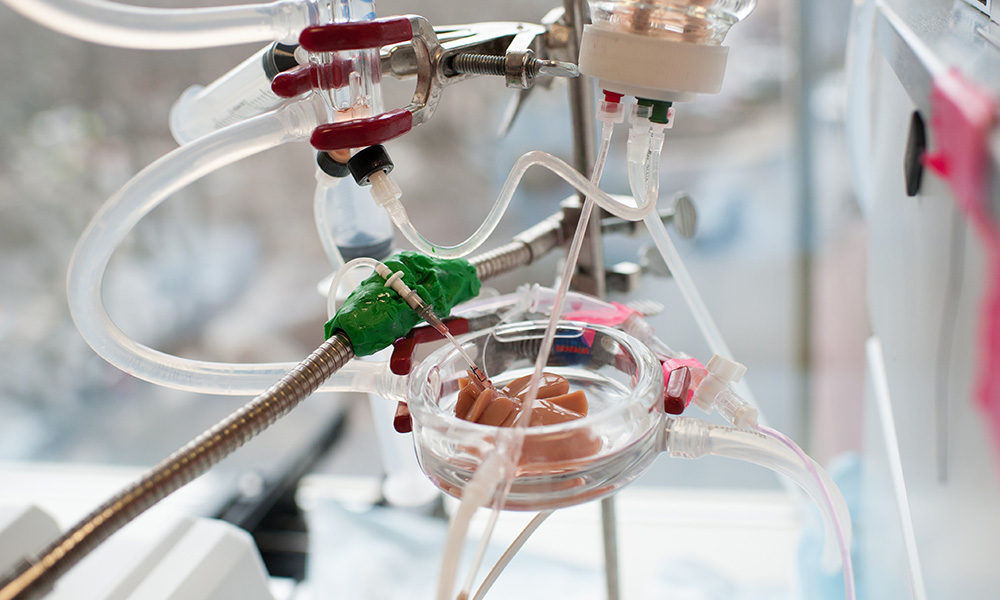
The CEMS is a place where scientific rigor and creativity are matched by a sense of sharing and community. The center draws strength from its broad and collaborative spirit and from its affiliations with surrounding biomedical research institutions including Massachusetts General Hospital, Harvard Medical School, Shriners Burns Hospital and Massachusetts Institute of Technology.
News
Recent news and upcoming events from the CEMS at Massachusetts General Hospital.
Our Team
Learn about the faculty, postdocs, technicians and other staff who make up our team.
Research Thrusts
The CEMS focuses on several different research thrusts that combine the principles of engineering and medicine in order to find new solutions that push the envelope of health care technology.
Education and Training
The CEMS provides education and training opportunities for postdoctoral and predoctoral students.
News and Announcements
About the CEMS
The Center for Engineering in Medicine & Surgery (CEMS) draws strength from its diversity and collaborative spirit, and from its affiliations with surrounding biomedical research institutions including the Massachusetts General Hospital, the Harvard Medical School, the Shriners Burns Hospital, and the Massachusetts Institute of Technology.