The Healey Center is partnering with Tackle ALS and several pharmaceutical companies to evaluate five therapies within one trial with the new HEALEY ALS Platform Trial.
The experimental treatments include: zilucoplan, a small macrocyclic peptide inhibitor of complement component 5 [C5], developed by Ra Pharmaceuticals, Inc.; Verdiperstat, an oral myeloperoxidase inhibitor, developed by Biohaven Pharmaceutical Holding Company Ltd.; Bioenergetic Nanocatalysis (CNM-Au8, nanocrystalline gold) developed by Clene Nanomedicine, Inc.; Pridopidine, a highly selective S1R agonist, developed by Prilenia Therapeutics; and IC14 immunotherapy, developed by Implicit Bioscience Ltd.
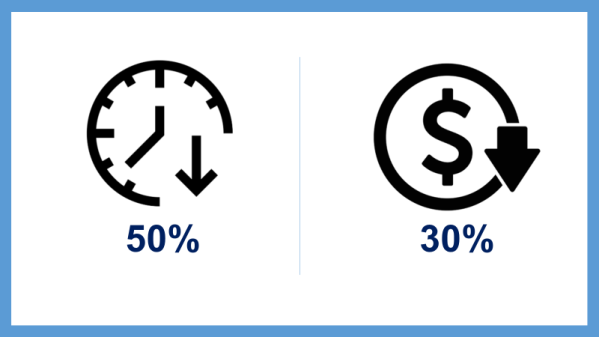
We worked closely with Berry Consultants, the leading statistical group for adaptive platform trials, and the Northeast ALS Consortium (NEALS), the largest ALS clinical trial network in the world, to design the HEALEY ALS Platform. The result is a truly revolutionary clinical trial approach that cuts the time to finding an effective treatment by 50 percent and decreases development costs by about 30 percent.
Importantly, the trial will dramatically increase the number of people with ALS on active treatment: in this new approach, 3 out of 4 participants are assigned to active study medication and, after six months, everyone who completes the trial has the option to receive active treatment. This trial allows us to add or drop therapies once their effectiveness is determined without the need to stop and restart the trial. Built-in interim early success/futility analyses further accelerate the time to first effective treatment by allowing identification of successful treatments rapidly while dropping ineffective treatments quickly from the platform.
 In addition to evaluating the effectiveness of therapeutics, the HEALEY ALS Platform Trial also provides the opportunity to identify new biomarkers, which help detect response to treatment faster than traditional ALS measures, and may allow us to diagnose the disease earlier and, ultimately, prevent ALS.
In addition to evaluating the effectiveness of therapeutics, the HEALEY ALS Platform Trial also provides the opportunity to identify new biomarkers, which help detect response to treatment faster than traditional ALS measures, and may allow us to diagnose the disease earlier and, ultimately, prevent ALS.
The trial will continue to adapt and incorporate new learnings so that future treatments will be tested in the platform using even more efficient biomarkers.
Enrollment in the first three therapy regimens will start in early 2020, and we expect to enroll 160 people in each for a total of 480 people to start. Fifty-four centers from across the United States will participate in the HEALEY ALS Platform Trial, ensuring close and broad access to people with ALS. Pending additional support, we plan to add the fourth treatment in Q 2 2020 and the fifth in Q 4 2020. We plan to continue adding more therapies to the platform each year.
With this tool, testing continues until cures are found. There is huge support and enthusiasm for this approach from people and families with ALS, the FDA and ALS neurologists. We could not have done this without your support. Thank you!
We look forward to providing you with updates on the progress of this work. If you would like to learn more about the HEALEY ALS Platform Trial and how you can participant in this effort please contact us at HealeyCenterforALS@mgh.harvard.edu.


 In addition to evaluating the effectiveness of therapeutics, the HEALEY ALS Platform Trial also provides the opportunity to identify new biomarkers, which help detect response to treatment faster than traditional ALS measures, and may allow us to diagnose the disease earlier and, ultimately, prevent ALS.
In addition to evaluating the effectiveness of therapeutics, the HEALEY ALS Platform Trial also provides the opportunity to identify new biomarkers, which help detect response to treatment faster than traditional ALS measures, and may allow us to diagnose the disease earlier and, ultimately, prevent ALS.